Unexpectedly attractive molecules
A π-stacked dimer of positively charged antiaromatic molecules stabilized upon stacking
An Ehime University research group has discovered that antiaromatic molecules, typically unstable, can form stable dimers through π-stacking. The homoHPHAC cation, despite its positive charge, adopts a slightly offset stacked structure. This interaction induces electronic reorganization, partially attenuating antiaromaticity. The findings reveal a new mode of molecular assembly for antiaromatic molecules and provide insights for designing functional π-conjugated materials.
Aromatic molecules are known for their stable electronic structures. These molecules typically adopt planar geometries with delocalized π-electrons and can form assemblies through π–π stacking interactions. In contrast, antiaromatic molecules, which possess fundamentally different electronic structures, are inherently unstable, and it has been considered that their instability can be alleviated when two molecules fully overlap.
A research group at Ehime University investigated a π-conjugated molecule they developed, the homoHPHAC cation, focusing on its molecular structure and aggregation behavior. They found that, upon partial removal of peripheral substituents, the molecule forms a π-stacked dimer with overlapping π-surfaces despite its positive charge.
X-ray crystallographic analysis revealed that the two molecules adopt a slightly offset “slip-stacked” arrangement rather than a perfectly face-to-face geometry. The interplanar distance is approximately 3.3 Å, which is typical of π–π interactions. Furthermore, NMR measurements in solution demonstrated a monomer–dimer equilibrium dependent on both concentration and temperature.
Furthermore, electronic structure analysis based on quantum chemical calculations using the crystal structure revealed that intermolecular stacking stabilizes the molecular orbitals, leading to partial attenuation of the intrinsic instability characteristic of antiaromatic molecules. These results suggest that the findings may extend the conventional understanding that stabilization of antiaromatic molecules occurs only through perfectly π-stacked dimer formation.
Experimental examples of such π-stacked dimers of antiaromatic molecules remain extremely rare, and this study significantly advances the understanding of their behavior. The findings are expected to contribute to the development of design principles for molecular assemblies, paving the way for new electronic materials and functional π-conjugated systems.
Reference URL: https://doi.org/10.1039/D5CC07392H
Bibliographic Information
Title: π-Stacked Dimerization of an Antiaromatic HomoHPHAC Monocation
Authors: Kaito Wada (Ehime Univ.), Yuma Tanioka (Ehime Univ.), Shigeki Mori (Ehime Univ.), Hidemitsu Uno (Ehime Univ.), and Masayoshi Takase* (Ehime Univ.) (*corresponding author)
Journal: Chemical Communications2026, 62, 6716–6720
DOI: 10.1039/D5CC07392H
Publication Date: March 4, 2026
Fundings
- Japan Society for the Promotion of Science (JSPS) KAKENHI (JP24K01470)
Media
-
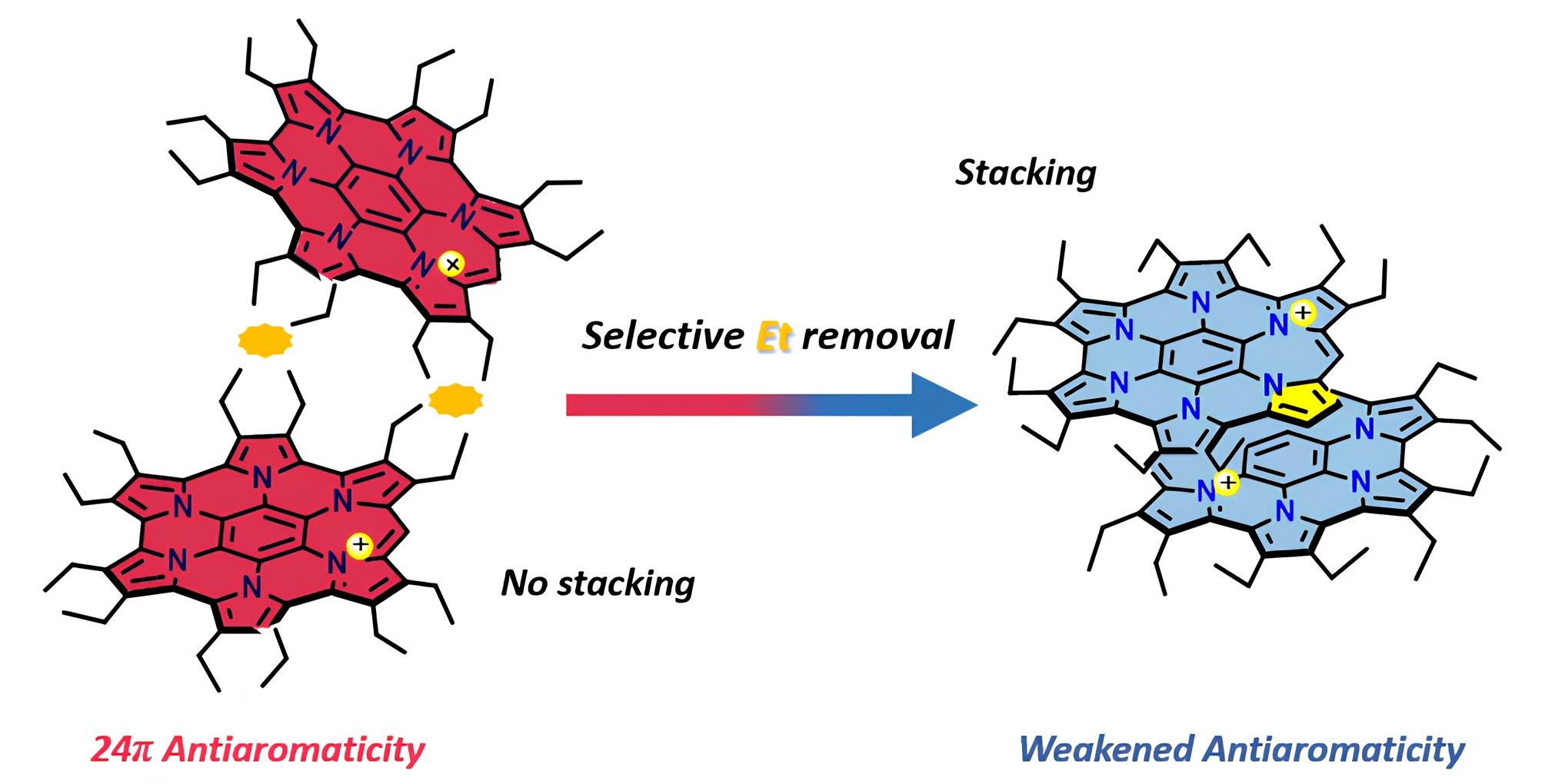
Selective substituent removal leads to π-stacked dimer formation of an antiaromatic monocation
Removal of peripheral substituents leads to the formation of a slightly offset “slip-stacked” structure of a positively charged antiaromatic molecule, resulting in a stabilized dimer with attenuated antiaromaticity.
credit : Royal Society of Chemistry
Usage Restriction : Please get copyright permission -

Harvesting substituents enables π-stacked dimers of antiaromatic cation
“Harvesting” peripheral substituents reveals a highly planar antiaromatic molecule (homoHPHAC cation). Despite electrostatic repulsion between positively charged species, the molecules adopt a slip-stacked arrangement, forming a stable π-stacked dimer. This stacking induces electronic reorganization, leading to partial attenuation of antiaromaticity. The terraced landscape in the background symbolizes the potential for creating new modes of molecular assembly through rational molecular design.
credit : Royal Society of Chemistry
Usage Restriction : Please get copyright permission
Contact Person
Name : Masayoshi Takase
Phone : +81-89-927-9610
E-mail : takase.masayoshi.ry@ehime-u.ac.jp
Affiliation : Graduate School of Science and Engineering, Ehime University
