Molecular identification of an enzyme reported over 60 years ago
Biochemical and structural biological analysis of a hypothetical gene (protein) within a gene cluster related to metabolic pathway of L-threonate, a four-carbon sugar acid, revealed it’s enzyme function as a L-threonate 3-dehydrogenase and physiological role for metabolic diversity. Although the existence of this enzyme itself was reported in the same academic journal in 1964, the molecular identity of which has remained unknown for over 60 years.
In the 1950s to 1970s, discovery of enzyme began by identifying new chemical reactions within cell-free extracts, but their molecular identification among (function unknown) hypothetical genes (proteins) is difficult.
Sugar acids are compounds formed by the oxidation of aldose monosaccharides. Metabolic genes for C4 and C5/C6 sugar acids are separately located on bacterial genomes. However, we discovered that in several bacteria, including the marine bacterium Paracoccus litorisediminis, these genes form a single cluster, in which a homologous gene to GL300_RS07945 was usually contained. GL300_RS07945, belonging to SDR protein superfamily, was a typical “function unknown gene” with less than 30% amino acid sequence similarity to any known functional protein.
In known metabolic pathway for L-threonate (Fig.1), the C4 sugar acid, L-threonate is converted to 3-oxo-L-threonate through sequential reactions by dehydrogenase (Ltn2D) and isomerase (OtnI). Substrate screening revealed that GL300_RS07945 could oxidize L-threonate, suggesting two enzymes with seemingly identical activities within the same gene cluster. Surprisingly, GL300_RS07945 was “L-threonate 3-dehydrogenase (Ltn3D),” which performs the reaction of Ltn2D/OtnI in a single step. Crystallographic analysis revealed that the positional relationship between the carbon atom at the 3-position of tartronic acid, a competitive inhibitor, and the nicotinamide ring of the coenzyme NADP+, perfectly matched the function of Ltn3D (Fig.2). Unlike Ltn2D, Ltn3D can oxidize not only L-threonate but also C5/C6 sugar acids, by which C5/C6 sugar acid enable to be metabolized via L-threonate pathway.
Although the existence (activity) of Ltn3D itself was reported in the same academic journal in 1964 and was even assigned an EC number (EC1.1.1.129), the molecular identity of which has remained unknown for over 60 years.
Bibliographic Information
Title: Identification, functional characterization, and structural analysis of an atypical L-threonate 3-dehydrogenase
Authors: Seiya Watanabe, Himika Sato, Taiyo Yokoi, Shin-ichi Terawaki
Journal: Journal of Biological Chemistry
DOI: 10.1016/j.jbc.2026.111280
Fundings
- Japan Society for the Promotion of Science (JSPS) KAKENHI 24K09366
Media
-
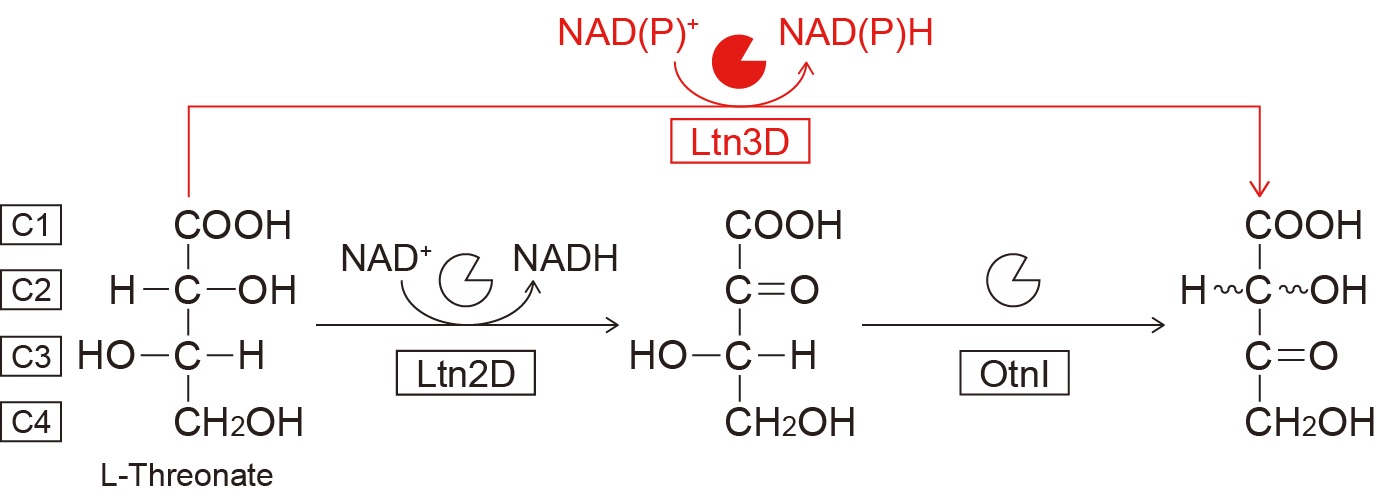
L-Threonate metabolism from bacteria
Ltn3D catalyzes one step reaction for oxidization of C3 hydroxyl group of L-threonate, differed from two steps by Ltn2D and OtnI involved in known L-threonate pathway.
credit : Seiya Watanabe
Usage Restriction : Please get copyright permission -
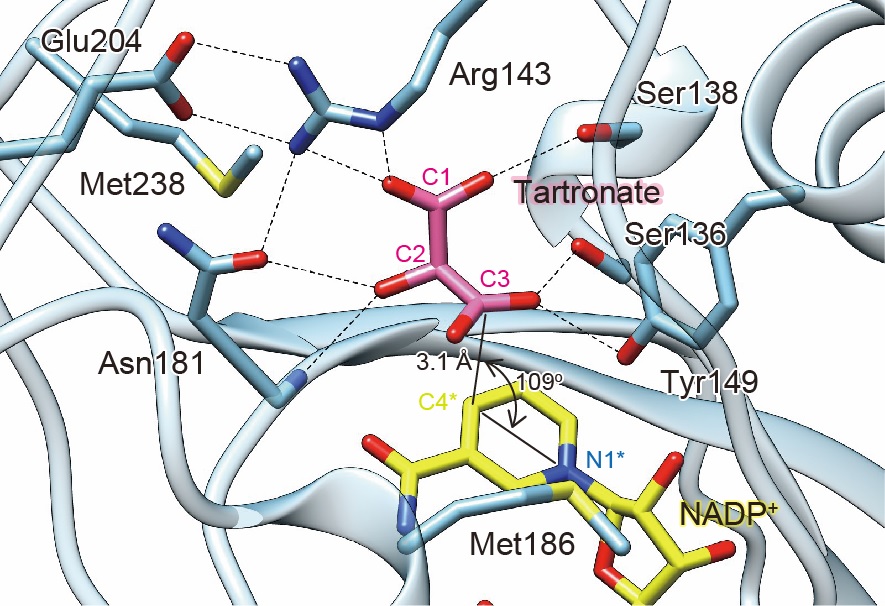
Crystal structure of Ltn3D in complex with coenzyme (NADP+) and tartronate (competitive inhibitor)
The distances and angle between tartronate and NADP+ were significantly favorable for the catalysis as Ltn3D.
credit : Seiya Watanabe
Usage Restriction : Please get copyright permission
Contact Person
Name : Seiya Watanabe
Phone : +81 89-946-9848
E-mail : irab@agr.ehime-u.ac.jp
Affiliation : Graduate School of Agriculture, Ehime University

